Downloads
DWR Diagnostics, based at DWR Veterinary Specialists, is a well-established laboratory providing a comprehensive range of diagnostic testing
Fine Needle Aspiration
The technique is not a strictly aspetic procedure, but it is wise to clip the area and swab with alcohol. The mass/lymph node is immobilised with one hand and a 23 ga needle +/- a 5 ml syringe is inserted into the lesion. The sample is obtained in 1 of 3 ways:
A. “Needle only” method
This method is useful for aspirating soft masses and lymph nodes and has the advantages that delicate, fragile cells are not damaged by suction and haemodilution is minimised.
- A 23 ga needle is inserted (without a syringe)
- The needle is moved to and fro’ within the mass, whilst redirecting it in many directions
- Care is taken to ensure the needle is not pushed through the mass into the surrounding tissues
- A sample is obtained within the bore/hub of the needle.
B. “Continuous suction” method
This method is useful for aspirating firm masses such as fibrosarcomas which tend not to exfoliate cells well.
- A 23 ga needle with a 5 ml syringe is inserted into the lesion
- Continuous suction is applied by withdrawing the plunger (3 – 5 mls of suction are sufficient)
- Whilst suction is maintained the needle is moved to and fro’ within the lesion, redirecting each time, again taking care to avoid pushing the needle through the mass into the surrounding tissues
- Suction is released before removing the needle.
C. “Intermittent suction” method
This method is suitable for small masses where it is not possible to redirect the needle without exiting the mass.
- A 23 ga needle and 5 or 10 ml syringe is inserted into the mass
- The plunger is withdrawn and released several times
- Suction is released before removing the needle from the lesion.


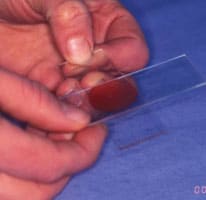
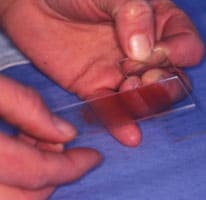
Making the Smear (see images above)
An air-filled syringe is attached to the needle (if the syringe was already attached to the needle it should be disconnected from needle, filled with air and then re-attached to the needle). Holding the tip of the needle over a clean glass slide, the plunger is briskly depressed, thus expelling the contents of the needle onto the slide (image 1 above) (now referred to as slide 1), or, if there is a high harvest, distributing between 2 or 3 slides laid side by side.The sample is then smeared using the squash preparation technique. NB it is very important to smear the sample rather than just squirting it on to a slide, since the cells must be in a monolayer to be evaluated.
A. Squash preparation
- Slide 1 is held in the left hand (for right handed people)
- A second side (now referred to as slide 2) held in the right hand and placed flat onto slide 1 at right angles to it
- This spreads the sample (image 2 and 3 above)
- If necessary gentle pressure may be applied using the right thumb to encourage the cells to spread out. Excessive pressure will result in cell rupture.
Slide 2 is slid quickly and smoothly across slide 1, (image 4 above)
The smear is on the lower surface of slide 2
With experience this technique produces excellent smears. Practice is required!
Smearing fluid samples
For turbid samples the blood smear technique is used (see preparing a blood film), or can be modified with the line concentration technique.
Line concentration technique
The sample is smeared as for the blood smear technique, but when the spreader slide has been advanced about two-thirds of the way across the slide the spreader slide is abruptly lifted upwards. This produces a smear with a concentrated line of cells at its end, instead of a feathered edge.
Impression smears
- Impression smears are made before and after cleaning the surface of the ulcerated mass with a saline-moistened swab and blotting dry
- A clean glass slide is gently touched to the lesion and immediately lifted off without smearing the slide across the lesion
- The procedure is repeated to obtain several imprints on one slide.
Impression smears may also be made from the cut surface of tissues obtained at surgery or post mortem. Blood and tissue fluid should be removed by blotting the sample dry before touching the sample onto a slide.
Scrapings
- The lesion is first cleaned and blotted dry
- A scalpel blade is scraped across the lesion’s surface several times
- Material collected on the blade is transferred to a slide by gently teasing it off the blade using a needle or the blade can be used like a paintbrush to gently smear the sample onto a slide
- If the material is thickly spread a squash preparation is made as described for fine needle aspirates.
Swab smears
A swab or cotton bud is used to collect samples from fistulous tracts and from the vagina. Unless the lesion is very moist the swab must first be moistened in isotonic saline. The swab is rubbed against the surface of the lesion and then gently rolled along a slide, taking care not to rub the swab across the slide since this would damage the cells.
